AP Chemistry Review – Work In Progress
AP Chemistry Review – Work In Progress
I think it will fun to write a review of AP Chemistry. This course is a great way to get a sense of how difficult general chemistry classes are at the college level.
Content covered on the AP Chemistry exam:
- Atomic Structure and Properties
- Molecular and Ionic Compound Structure and Properties
- Intermolecular Forces and Properties
- Chemical Reactions
- Kinetics
- Thermodynamics
- Equilibrium
- Acids and Bases
- Applications of Thermodynamics
Atomic Structure and Properties
This unit covers a lot of the properties of elements on the periodic table.
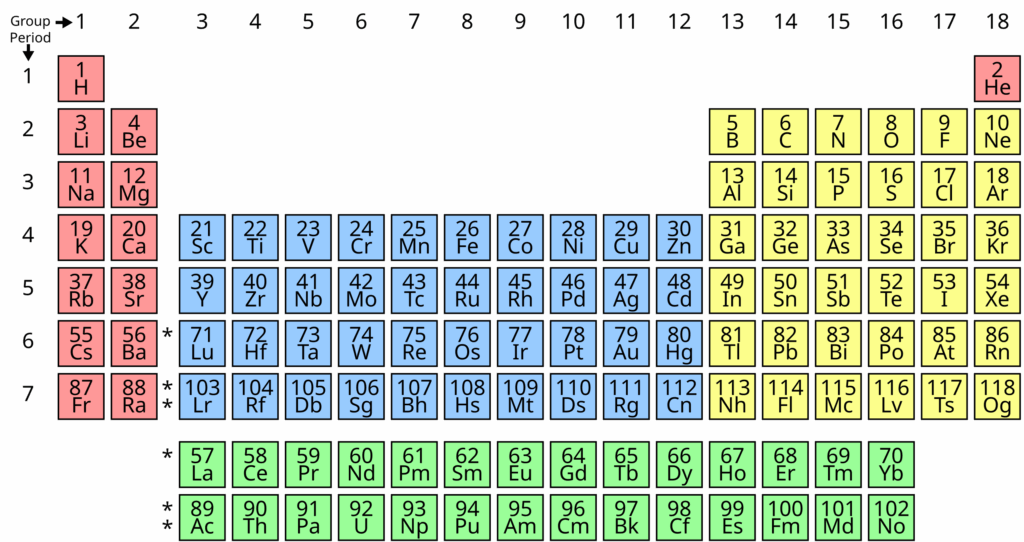
It is important to know to read the periodic table.
Identifying the atomic number, symbol, and atomic mass from the periodic table of element is an important skill.
It will also be important to memorize properties like Avogadro’s number: 6.022 * 10^23 atoms/mol.
Moles(mol) are chemistry units. As an example, there are 6.022 * 10^23 atoms of Carbon per mole of Carbon. Carbon has an atomic mass of 12.01.
You could probably find this on another periodic table that lists the atomic mass of the elements. In terms of the atomic mass of Carbon, you would need 12.01 grams of Carbon to make 1 mole of Carbon. That 1 mole of Carbon would have 6.022 * 10^23 atoms of Carbon.
In terms of mass, we used grams in this case.
Metric Unit Prefixes
For metric units, we can use prefixes to help specify the amount of the unit. For example, 1 kilogram is equal to 1000 grams. We might write kilogram as kg and grams as g.
| Factor | Prefix | Symbol |
| 10^9 | giga | |
| 10^6 | mega | |
| 10^3 | kilo | |
| 10^-2 | centi | |



